1Department of Laboratory Medicine, Seoul Medical Center, Seoul, Korea
2Department of Laboratory Medicine and Genetics, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea
3Department of Laboratory Medicine, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea
4Department of Medical Device Management and Research, Samsung Advanced Institute for Health Sciences & Technology, Sungkyunkwan University, Seoul, Korea
Correspondence to Tae Yeul Kim E-mail: eilpost@skku.edu; Eun Jeong Won E-mail: ejwon@amc.seoul.kr
Ann Clin Microbiol 2026;29(2):6. https://doi.org/10.5145/ACM.2026.29.2.6
Received on 26 February 2026, Revised on 14 April 2026, Accepted on 7 May 2026, Published on 8 May 2026.
Copyright © Korean Society of Clinical Microbiology.
This is an Open Access article which is freely available under the Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License (CC BY-NC-ND) (https://creativecommons.org/licenses/by-nc-nd/4.0/).
Background: To overcome the labor-intensive and technician-dependent nature of the formalin–ether concentration technique (FECT), artificial intelligence (AI)-based automated feces analyzers have been developed for detecting fecal parasites. This study compared the clinical performance of the KU-F40 AI-based automated system with that of FECT in a routine diagnostic environment.
Methods: In total, 1,011 fecal samples were prospectively collected and analyzed concurrently via both FECT and the KU-F40 system. The diagnostic performance of the KU-F40 system was evaluated using FECT as the reference method. The clinical performance parameters included accuracy, Cohen’s kappa coefficient, sensitivity, specificity, positive predictive value (PPV), and negative predictive value (NPV).
Results: The KU-F40 system exhibited an accuracy of 98.5% and Cohen’s kappa of 0.608 relative to FECT. The sensitivity and specificity of the KU-F40 system were 57.1% (95% confidence interval [CI], 34.0–78.2) and 99.4% (95% CI, 98.7–99.8), respectively. When adjusted for an institutional prevalence of 0.45%, its PPV and NPV were 29.9% (95% CI, 15.0–50.7) and 99.8% (95% CI, 99.7–99.9), respectively. Among the 27 parasite-positive samples identified using at least one method, the methods achieved concordant species identification for six samples (22.2%). The KU-F40 system offers superior workflow efficiency by reducing labor time and hazard exposure.
Conclusion: The KU-F40 system demonstrated high specificity and negative predictive value in a low-prevalence setting; however, its limited sensitivity indicates it should not replace conventional examination when clinical suspicion is high. Despite these limitations, it may serve as a useful adjunctive tool that improves laboratory workflow efficiency and minimizes exposure to hazardous reagents.
Artificial intelligence, Automated feces analyzer, Predictive value of tests, Intestinal parasites, Republic of Korea
Intestinal parasitic infection is an important public health problem worldwide [1]. In the Republic of Korea, the overall prevalence of parasitic infections has decreased dramatically since the 1970s [2]. However, specific parasites, particularly Clonorchis sinensis, remain endemic in riverside areas and pose a continuous public health challenge owing to their carcinogenic potential [3,4]. Imported parasitic diseases are increasingly being reported [5]. The formalin–ether concentration technique (FECT) method is the routine reference standard for detecting and identifying the microscopic ova and parasites present in such infections [6]. However, this method requires the use of hazardous reagents and is labor-intensive, often leading to low sensitivity and inter-observer variability owing to its high dependence on technical proficiency [7].
To overcome these limitations, various approaches, ranging from modified concentration techniques to fully automated systems, have been explored [8,9]. Recently, artificial intelligence (AI)-assisted image-based analyzers have been developed to address the workflow challenges of conventional microscopy [10,11]. Although these systems are expected to improve laboratory efficiency, reduce operator fatigue, improve biosafety, and standardize the screening process, the data on their clinical performance in routine diagnostic environments remains limited. Consequently, studies that evaluate a broad spectrum of parasites, including the ova and protozoa commonly encountered in South Korean clinical laboratories, are urgently required to validate the practical utility and diagnostic accuracy of these AI-based automated systems.
To address this need, this study evaluated the clinical performance of the KU-F40 AI-based automated fecal analyzer (Zhuhai Keyu Biological Engineering) for prospectively collected fecal samples, comparing the results with those of FECT.
This single-center prospective study was conducted at the Clinical Microbiology Laboratory of the Samsung Medical Center, Seoul, South Korea, from April 2025 to July 2025. This study evaluated the clinical performance of the KU-F40 automated analyzer, using FECT as a reference method. The analyses were performed concurrently by technicians blinded to the results.
We collected data from fecal samples ordered for routine ova and parasite examination during the study period. Only samples that underwent both tests (FECT and KU-F40 automated analysis) were included. Consequently, 1,011 samples were included in the final analysis. Repeated samples from the same patient during the study period were excluded.
The KU-F40 system integrates high-resolution digital microscopy with AI recognition algorithms to analyze the elements formed. For each automated test, approximately 1 g of fresh fecal specimen was collected in a dedicated collection cup. The analyzer automatically performs quantitative dilution, mixing, and mechanical homogenization using a proprietary dilution solution. The resulting suspension was filtered and introduced into a two-channel flow counting cell, with a 100 μL capacity per cell.
Following a 45 s sedimentation period, a five-megapixel complementary metal-oxide-semiconductor (CMOS) camera was used to capture images under the manufacturer-recommended settings: in low-power mode (20× magnification), for helminth egg detection, images were taken across a single focal layer (1,560 pieces/layer), whereas in high-power mode (40× magnification), for protozoa detection, 20 fields were captured across two focal layers per field. The integrated AI algorithm performed preliminary identification of parasites and other elements. Before final verification, all images from each sample were saved and reviewed by laboratory personnel to rule out misidentification caused by debris.
Conventional examination of ova and parasites was performed following standard laboratory protocols. Fecal samples were processed using a Mini Parasep device (Apacor) to determine the formalin–ether concentration. Ethyl acetate (1.0 mL) was then added to the mixing chamber. Approximately 2 g of fecal sample was collected using a spoon at the end of the filter and sealed in a tube. The chamber was vortexed to emulsify the specimen and then centrifuged at 1,200 ×g for 3 min. After centrifugation, the mixing chamber and filter were discarded. The supernatant was decanted, and 1–2 drops of physiological saline were added to the remaining sediment. The sediment was homogenized by vortexing, and slides were prepared for microscopic examination. Skilled technicians screened the entire field at 100× magnification, with further confirmation of helminths or protozoa at 400× magnification.
The diagnostic accuracy of the KU-F40 system and the agreement between the FECT and KU-F40 systems were assessed by calculating the overall accuracy and Cohen’s kappa coefficient. The diagnostic performance of the KU-F40 system was evaluated using FECT as the reference standard. Sensitivity, specificity, positive predictive value (PPV), and negative predictive value (NPV) were calculated accordingly.
To reflect clinical utility, PPV and NPV were estimated by adjusting for an institutional prevalence of 0.45%. The institutional prevalence was estimated from the laboratory’s annual surveillance data from September 2024 to August 2025, which included but was not limited to the study period. The species identification concordance rate was calculated for all samples identified as positive for parasitic infection by at least one method. Statistical analyses were performed using R 4.4.2.
To assess the performance of the KU-F40 automated system, its accuracy and agreement with FECT were analyzed for 1,011 specimens (Table 1). The KU-F40 system achieved a high overall accuracy of 98.5% (95% confidence interval [CI], 97.5–99.2), with a Cohen’s kappa of 0.608 (95% CI, 0.426–0.790), indicating moderate to substantial agreement between the manual and automated workflows. The KU-F40 system exhibited a diagnostic sensitivity of 57.1% (95% CI, 34.0–78.2) and specificity of 99.4% (95% CI, 98.7–99.8). To evaluate the KU-F40 system’s clinical utility in a real-world setting, the predictive values were adjusted for a local institutional prevalence of 0.45%; under this low-prevalence condition, the PPV and NPV were 29.9% (95% CI, 15.0–50.7) and 99.8% (95% CI, 99.7–99.9), respectively.
Table 1. Diagnostic performance of the KU-F40 system, using FECT as the reference method
| KU-F40 system | ||||
|---|---|---|---|---|
| Positive | Negative | Total | ||
| FECT | Positive | 12 | 9 | 21 |
| Negative | 6 | 984 | 990 | |
| Total | 18 | 993 | 1,011 | |
| Accuracy: 98.5% (95% CI, 97.5–99.2) Sensitivity: 57.1% (95% CI, 34.0–78.2) Specificity: 99.4% (95% CI, 98.7–99.8) PPVa): 29.9% (95% CI, 15.0–50.7) NPVa): 99.8% (95% CI, 99.7–99.9) | ||||
a)PPV and NPV were adjusted based on an institutional prevalence of 0.45%.
Abbreviations: FECT, formalin–ether concentration technique; CI, confidence interval; PPV, positive predictive value; NPV, negative predictive value.
Within the 21 samples identified via FECT as positive for parasitic infection, the most prevalent parasites were Metagonimus yokogawai (n = 4 samples) and Trichuris trichiura (n = 4), followed by C. sinensis (n = 3), Endolimax nana (n = 3), and Entamoeba coli (n = 3). Among the 12 samples that were positive by both methods, species-level concordance was observed in 6 (50.0%) cases. When considering all 27 samples positive by at least one method, complete agreement in both detection and species identification was 6 (22.2%) (Table 2). Trematode identification varied notably between the methods: four cases reported as M. yokogawai by FECT were identified as C. sinensis by the KU-F40 system, with detection counts ranging from 1 to 26 eggs per sample. Significantly, the KU-F40 system identified six cases of C. sinensis that were not detected by manual FECT. In all six instances, the automated system identified only a single egg per sample, representing the detection of low-density infections. Conversely, nine FECT-positive cases were not detected by the KU-F40 system: T. trichiura (n = 3), E. coli (n = 2), and Entamoeba dispar, C. sinensis, Blastocystis hominis, and Gymnophalloides seoi (n = 1 each); subsequent image review confirmed that none of these parasites or eggs were captured by the KU-F40 system. Fig. 1 presents representative images of the discordant cases captured using the KU-F40 system.
Table 2. Parasitic species identification using FECT and the KU-F40 system
| Concordance | Species identification | No. of cases | |
|---|---|---|---|
| FECT | KU-F40 system | ||
| Concordant | Endolimax nana | 3 | |
| Clonorchis sinensis | 2 | ||
| Entamoeba coli | 1 | ||
| Discordant | Metagonimus yokogawai | Clonorchis sinensis (Fig. 1A) | 4 |
| Trichuris trichiura | Ascaris lumbricoides (Fig. 1B) | 1 | |
| Entamoeba histolytica | Blastocystis hominis (Fig. 1C) | 1 | |
| Entamoeba coli (Fig. 1D) | |||
| Trichuris trichiura | Not detected | 3 | |
| Entamoeba coli | Not detected | 2 | |
| Entamoeba dispar | Not detected | 1 | |
| Clonorchis sinensis | Not detected | 1 | |
| Blastocystis hominis | Not detected | 1 | |
| Gymnophalloides seoi | Not detected | 1 | |
| Not detected | Clonorchis sinensis (Fig. 1E) | 6 | |
| Concordance rate | 22.2% (6/27) | ||
Abbreviation: FECT, formalin–ether concentration technique.
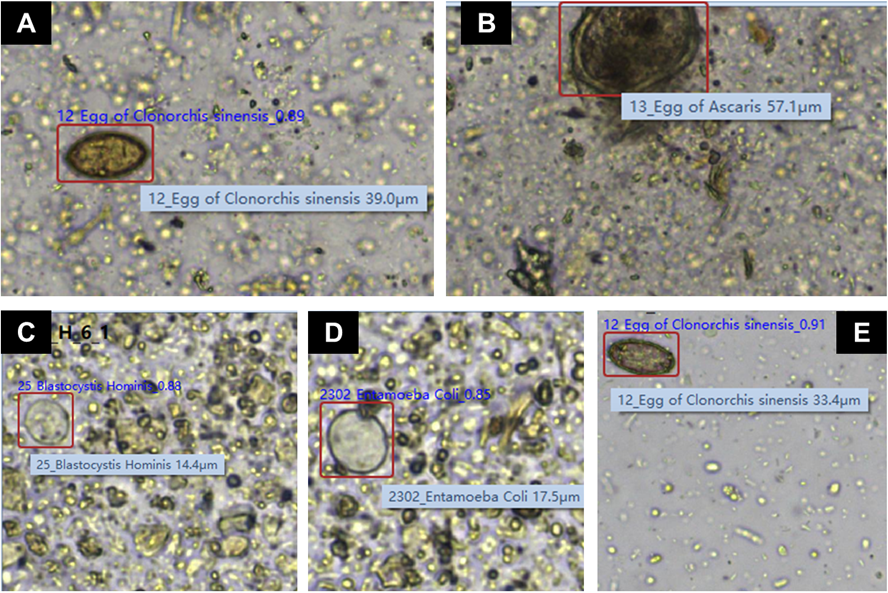
Fig. 1. Representative images, captured by the KU-F40 system, of the organisms identified differently by the FECT system. (A) Metagonimus yokogawai (FECT) vs. Clonorchis sinensis (KU-F40); (B) Trichuris trichiura (FECT) vs. Ascaris lumbricoides (KU-F40); (C) Entamoeba histolytica (FECT) vs. Blastocystis hominis (KU-F40); (D) Entamoeba histolytica (FECT) vs. Entamoeba coli (KU-F40); (E) Not detected (FECT) vs. Clonorchis sinensis (KU-F40). Images (A), (B), and (E) were captured using a 20× objective lens, while images (C) and (D) were captured using a 40× objective lens. FECT, formalin–ether concentration technique.
The operational efficiencies and technical profiles of the two methods were compared based on our institution’s average daily workload of 180 samples (Table 3). For this workload, the KU-F40 system required only 1.2 h of hands-on time, significantly less than the 7 h required for manual FECT.
Table 3. Workflow and technical characteristics of FECT and the KU-F40 systems
| Category | FECT | KU-F40 system |
|---|---|---|
| Analysis method | Manual microscopic examination | Automated image analysis |
| Operation mode | Manual | Automated |
| System configuration | Open system | Closed system |
| Chemical exposure | Frequent (Formalin, ethyl acetate) | Minimal (Automated processing) |
| Hands-on time (180 samples/daya)) | 7 h | 1.2 h |
| Digital records | Not available | Available (Images) |
| Mean TAT (per test) | 2.3 minb) | Variablec) |
a)Based on the average daily workload at our institution; b)Mean TAT per test based on a daily batch of 180 samples; c)With the high power setting fixed at 20 fields/2 layers, the TAT of the KU-F40 system varies according to the lowpower settings. The following TATs were provided by the manufacturer: 2.9 min/test (at 400 pieces/layer), 3.9 min/ test (at 584 pieces/layer), 9.6 min/test (at 1,560 pieces/layer).
Abbreviations: FECT, formalin–ether concentration technique; TAT, turnaround time.
In terms of safety, KU-F40 functions as a closed and automated system, thus minimizing the risk of exposure to biological and chemical hazards. In contrast, manual FECT operates as an open system, requiring frequent specimen handling and potential exposure to hazardous reagents such as formalin and ethyl acetate. The KU-F40 system supports archiving of digital image records for all samples, a feature not available with manual FECT. The mean turnaround time (TAT) for FECT was 2.3 min per test, whereas for the KU-F40 system, mean TAT varied depending on the scan settings.
This study evaluated the performance of the KU-F40 automated analyzer in a routine clinical setting, revealing moderate to substantial agreement (Cohen’s kappa, 0.608) between the KU-F40 and FECT results. Notably, the KU-F40 system exhibited high specificity (99.4%) and NPV (99.8%), confirming its potential to effectively rule out negative samples from low-prevalence populations. However, the system exhibited a relatively low sensitivity of 57.1%, and its PPV was only 29.9% (adjusted for our institutional prevalence of 0.45%). These findings suggest that while the KU-F40 system may be useful as an adjunctive workflow tool in low-prevalence laboratories, it should not replace FECT when high diagnostic sensitivity or precise species-level identification is required.
FECT achieved an overall infection detection rate of 2.08% (21/1,011), slightly higher than the rate of 1.78% (18/1,011) achieved by the KU-F40 system. In contrast, Huang et al. [10] found that an automated system achieved a significantly higher detection rate (8.74%) than manual microscopy (2.81%). This discrepancy likely stems from the fact that Huang et al. [10] used routine direct saline smears, a method that does not involve concentration of the samples and that uses significantly smaller sample volumes than FECT. Here, however, we utilized FECT as a more rigorous reference standard. Consequently, our results are more consistent with those of Boonyong et al. [12], who reported that FECT identified more positive samples than an automated analyzer (468 vs. 440 positive cases, respectively, for FECT and the automated system, among 800 preserved samples) [12]. This disparity is primarily because the manual concentration procedure of FECT uses a larger stool volume than the automated analyzer and because FECT effectively removes fat and debris using ethyl acetate. In summary, the KU-F40 system provides as a practical solution to bridge the gap between traditional methods. While routine direct smear analysis is convenient, it carries a high risk of false negatives. Although FECT provides superior sensitivity, its high labor intensity and risk of exposure to hazardous reagents present significant drawbacks.
The operational efficiency of the KU-F40 system was evaluated alongside that of FECT. The KU-F40 system reduced hands-on labor time and chemical exposure, although its total instrument turnaround time varied according to scan settings and was not necessarily shorter than FECT. In clinical laboratories, the KU-F40 system may complement traditional methods. Here, while FECT identified 21 positive cases, a total of 27 samples were positive by at least one of the two methods. Our findings reveal that the KU-F40 system can identify low-density positive cases, such as those with low-density C. sinensis infection, that may be overlooked during manual FECT. Considering that C. sinensis is the most prevalent and medically important parasite in South Korea, the ability of this system to detect low-density infections is of paramount clinical significance.
The system may have advantages for detecting certain helminth eggs, particularly when images are captured at 20× magnification, but species-specific sensitivity could not be estimated reliably because of the small number of positive samples. This potential benefit likely stems from the multilayer scanning protocol which mimics manual fine focusing [13]. While 20× magnification is sufficient to recognize the relatively large and distinct morphological features of helminth eggs, it limits the fine differentiation required for identifying and distinguishing between small and similar protozoa [10,13]. In settings with a high prevalence of protozoa, adjusting the parameters to acquire more images at 40× magnification may improve detection. Further studies across diverse settings are required to determine the optimal imaging parameters. Our finding of exceptionally low species-level concordance (only six cases) suggests that the AI training database requires further expansion and refinement. Furthermore, owing to the limited resolution and image quality, the objects in the images exhibit ambiguous or atypical morphologies. Such suboptimal imaging leads to an interpretive gap between automated categorization and expert review, potentially challenging even experts in performing accurate diagnosis. Therefore, while the KU-F40 system requires expert validation to reach its full potential, expert validation cannot fully compensate for the diagnostic uncertainty caused by suboptimal image quality.
Our study has several limitations. First, it was conducted at a single tertiary center in an urban area, where the institutional prevalence was 0.45%, and where most of the patients undergoing parasite testing were asymptomatic individuals undergoing routine health checkups. This geographic bias limited the number of positive specimens, and the study population may not exhibit the higher prevalence or epidemiological profiles of rural or riverside populations in South Korea. Second, the use of a localized study sample limited the diversity of parasite species in the samples. Although various species were identified, most of the positive cases exhibited one of a few prevalent species, such as M. yokogawai. Species endemic to specific regions, such as G. seoi, which is commonly found in river basins, were underrepresented. Therefore, the KU-F40 system’s performance for a broader spectrum of clinically significant helminths or protozoa could not be extensively evaluated. Future multicenter studies across diverse geographical regions are essential to provide the necessary datasets for further AI algorithm training and to validate the KU-F40 system’s performance in various epidemiological contexts. Third, FECT was used as the routine reference standard, although it is operator-dependent and may not represent an error-free gold standard. Unlike the KU-F40 system, in which high-resolution digital images are archived for retrospective review, manual FECT slides cannot be reverified in cases of discrepancy. This inability to confirm the discordant results from manual examination may have led to a conservative estimation of the diagnostic performance of the KU-F40 system. Further, the presence of ambiguous or atypical morphology in images captured by the KU-F40 system can challenge expert review and confirmation, particularly when only a single image is available. Here, for instance, an organism identified as Ascaris lumbricoides by the KU-F40 system was found to be as T. trichiura based on FECT. This case highlights the need for confirmatory testing via manual methods such as FECT or via molecular methods when image review is inconclusive.
The KU-F40 automated feces analyzer showed high specificity and a high NPV compared with FECT in a low-prevalence routine clinical laboratory setting. However, its limited sensitivity and low specieslevel concordance indicate that it should not replace conventional parasitological examination when clinical suspicion is high or when accurate species identification is required. The KU-F40 system may be useful as an adjunctive workflow tool that reduces hands-on time, supports digital image archiving, and minimizes laboratory exposure to hazardous reagents. Further multicenter studies using diverse parasite-positive samples and confirmatory methods for discordant results are needed to define its optimal clinical role.
The study protocol was approved by the Institutional Review Board (IRB) of the Samsung Medical Center (IRB No. 2026-01-174). The requirement for informed consent was waived as this study used anonymized data from residual diagnostic samples, which posed no risk to the subjects, and all data were processed to be untraceable.
The reagents used were provided by DIAGENEX Inc. Hee Jae Huh has been an editorial board member of the Annals of Clinical Microbiology since September 2024. However, she was not involved in the review process of this article. No potential conflicts of interest relevant to this article were reported.
This study was supported by a 2025 research grant from the Korean Society of Clinical Microbiology.
The datasets generated during the current study are available from the corresponding author upon request.
The authors are grateful to the members of the Tropical Medicine Study Group of the Korean Society of Clinical Microbiology for their valuable contributions to this study.
1. World Health Organization. Soil-transmitted helminth infections. https://www.who.int/news-room/fact-sheets/detail/soil-transmitted-helminth-infections [Online] (last visited on 26 December 2025).
2. Cho SY and Hong ST. What was the main factor in successful control of ascariasis in Korea? Parasites Hosts Dis 2023;61:103-26.


3. Jeong YI, Shin HE, Lee SE, Cheun HI, Ju JW, Kim JY, et al. Prevalence of Clonorchis sinensis infection among residents along 5 major rivers in the Republic of Korea. Korean J Parasitol 2016;54:215-9.


4. Sripa B, Bethony JM, Sithithaworn P, Kaewkes S, Mairiang E, Loukas A, et al. Opisthorchiasis and Opisthorchis-associated cholangiocarcinoma in Thailand and Laos. Acta Trop 2011;120:S158-68.


5. Chai JY. Imported parasitic diseases in the Republic of Korea: status and issues. J Korean Med Assoc 2025;68:52-76.
6. Garcia LS and Procop GW. Diagnostic medical parasitology. In: McPherson RA and Pincus MR, eds. Manual of Commercial Methods in Clinical Microbiology. 2nd ed. Washington, DC; ASM Press, 2016:284-308.

7. Ndao M. Diagnosis of parasitic diseases: old and new approaches. Interdiscip Perspect Infect Dis 2009;2009:278246.


8. Won EJ, Kim J, Ryang DW. Evaluation of modified formalin-ether concentration method using Para Tube in clinical settings. Ann Lab Med 2015;35:445-8.


9. Lee YJ, Won EJ, Cho YC, Kim SH, Shin MG, Shin JH. Utility of an automatic vision-based examination system (AVE-562) for the detection of Clonorchis sinensis eggs in stool. Ann Lab Med 2021;41:221-4.


10. Huang C, Huang Y, Lin F, Liao L. The clinical application value of the KU-F40 fully automatic fecal analyzer for the detection of fecal parasites: a large-sample retrospective study. Sci Rep 2025;15:24172.


11. Corpuz KDF, Kusolsuk T, Wongphan B, Chonsawat P, Naing KM, Boonsang S, et al. Performance validation of deep-learning-based approach in stool examination. Parasit Vectors 2025;18:322.


12. Boonyong S, Hunnangkul S, Vijit S, Wattano S, Tantayapirak P, Loymek S, et al. High-throughput detection of parasites and ova in stool using the fully automatic digital feces analyzer, orienter model fa280. Parasit Vectors 2024;17:13.


13. Aupaix A, Filippin L, Jaumot J, Cannoot S, Chemais M, Martiny D, et al. Evaluation of KU-F40 automated microscope for parasitology: when artificial intelligence meets old school microscopy. J Clin Microbiol 2026;64:e0074325.


1. World Health Organization. Soil-transmitted helminth infections. https://www.who.int/news-room/fact-sheets/detail/soil-transmitted-helminth-infections [Online] (last visited on 26 December 2025).
2. Cho SY and Hong ST. What was the main factor in successful control of ascariasis in Korea? Parasites Hosts Dis 2023;61:103-26.


3. Jeong YI, Shin HE, Lee SE, Cheun HI, Ju JW, Kim JY, et al. Prevalence of Clonorchis sinensis infection among residents along 5 major rivers in the Republic of Korea. Korean J Parasitol 2016;54:215-9.


4. Sripa B, Bethony JM, Sithithaworn P, Kaewkes S, Mairiang E, Loukas A, et al. Opisthorchiasis and Opisthorchis-associated cholangiocarcinoma in Thailand and Laos. Acta Trop 2011;120:S158-68.


5. Chai JY. Imported parasitic diseases in the Republic of Korea: status and issues. J Korean Med Assoc 2025;68:52-76.
6. Garcia LS and Procop GW. Diagnostic medical parasitology. In: McPherson RA and Pincus MR, eds. Manual of Commercial Methods in Clinical Microbiology. 2nd ed. Washington, DC; ASM Press, 2016:284-308.

7. Ndao M. Diagnosis of parasitic diseases: old and new approaches. Interdiscip Perspect Infect Dis 2009;2009:278246.


8. Won EJ, Kim J, Ryang DW. Evaluation of modified formalin-ether concentration method using Para Tube in clinical settings. Ann Lab Med 2015;35:445-8.


9. Lee YJ, Won EJ, Cho YC, Kim SH, Shin MG, Shin JH. Utility of an automatic vision-based examination system (AVE-562) for the detection of Clonorchis sinensis eggs in stool. Ann Lab Med 2021;41:221-4.


10. Huang C, Huang Y, Lin F, Liao L. The clinical application value of the KU-F40 fully automatic fecal analyzer for the detection of fecal parasites: a large-sample retrospective study. Sci Rep 2025;15:24172.


11. Corpuz KDF, Kusolsuk T, Wongphan B, Chonsawat P, Naing KM, Boonsang S, et al. Performance validation of deep-learning-based approach in stool examination. Parasit Vectors 2025;18:322.


12. Boonyong S, Hunnangkul S, Vijit S, Wattano S, Tantayapirak P, Loymek S, et al. High-throughput detection of parasites and ova in stool using the fully automatic digital feces analyzer, orienter model fa280. Parasit Vectors 2024;17:13.


13. Aupaix A, Filippin L, Jaumot J, Cannoot S, Chemais M, Martiny D, et al. Evaluation of KU-F40 automated microscope for parasitology: when artificial intelligence meets old school microscopy. J Clin Microbiol 2026;64:e0074325.

