1Institute of Medical Science, Gyeongsang National University, Jinju, Korea
2Department of Laboratory Medicine, GC Labs, Yangsan, Korea
Correspondence to Sunjoo Kim E-mail: sjkim8239@hanmail.net
Ann Clin Microbiol 2026;29(1):3. https://doi.org/10.5145/ACM.2026.29.1.3
Received on 8 January 2026, Revised on 4 February 2026, Accepted on 18 February 2026, Published on 9 March 2026.
Copyright © Korean Society of Clinical Microbiology.
This is an Open Access article which is freely available under the Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License (CC BY-NC-ND) (https://creativecommons.org/licenses/by-nc-nd/4.0/).
Background: Environmental contamination with multidrug-resistant organisms (MDROs), including carbapenem-resistant Acinetobacter baumannii (CRAB) and multidrug-resistant Pseudomonas aeruginosa (MRPA), remains a major challenge in healthcare facilities. Hypochlorous acid water (HOCl) has emerged as a promising disinfectant owing to its strong antimicrobial activity and favorable safety profile. This study aimed to evaluate the bactericidal efficacy of atomized HOCl against CRAB and MRPA in a hospital room.
Methods: An atomization experiment was conducted in a two-bed room. CRAB and MRPA were prepared using drying and non-drying methods, respectively. HOCl (CLFine) at concentration of 40 and 300 ppm was atomized using ultrasonic humidifiers. Bacterial samples were collected at 0, 1, 3, and 5 h after atomization. Viable bacterial counts were determined by culture, and bactericidal efficacy was evaluated.
Results: Atomized HOCl exhibited time- and concentration-dependent bactericidal effects against CRAB and MRPA. CRAB and MRPA reached their limits of detection at 3 and 5 h post-atomization at 40 ppm, and at 1 and 3 h at 300 ppm, respectively.
Conclusion: Atomized HOCl effectively inactivated CRAB and MRPA in a hospital room within 3–5 h. These findings support the potential application of HOCl atomization as an adjunctive environmental disinfection strategy for controlling MDRO contamination in healthcare facilities.
Acinetobacter baumannii, Cross infection, Disinfection, Hypochlorous acid, Pseudomonas aeruginosa
The global increase in multidrug-resistant organisms (MDROs) poses a serious threat to patient safety and healthcare systems worldwide. Among these pathogens, carbapenem-resistant Acinetobacter baumannii (CRAB) and multidrug-resistant Pseudomonas aeruginosa (MRPA) are recognized as particularly problematic causes of healthcare-associated infections, especially in intensive care units (ICUs) and critically ill or long-term hospitalized patients [1–4]. These organisms are frequently linked to ventilator-associated pneumonia, bloodstream infections, surgical site infections, and device-related infections and, therefore, to limited therapeutic options and increased mortality.
A. baumannii is of special concern because of its remarkable ability to survive for prolonged periods on dry surfaces, medical equipment, and hospital furnishings. Numerous investigations have demonstrated that hospital environments such as bed rails, ventilators, floors, and sinks act as persistent reservoirs for CRAB, contributing to sustained transmission and outbreak propagation despite routine cleaning [3–7]. Environmental persistence also plays a critical role in the transmission of P. aeruginosa, which is a highly adaptable opportunistic pathogen with intrinsic resistance to multiple antimicrobial classes and a strong capacity to acquire additional antimicrobial resistance. MRPA has increasingly been reported in healthcare-associated infections, and environmental reservoirs, including water systems, sinks, and moist surfaces, play a crucial role in its persistence within healthcare facilities [1,2,8]. Therefore, environmental contaminants are key targets in infection prevention and control strategies.
Conventional disinfection practices largely rely on manual surface cleaning using liquid disinfectants, which may fail to achieve uniform decontamination in complex hospital environments. The effectiveness of these approaches is highly dependent on the operator’s technique, workload, and compliance, potentially leaving uncleaned areas that permit microbial survival [8].
Hypochlorous acid water (HOCl), a chlorine-based disinfectant generated by electrolysis, has attracted considerable interest because of its broad-spectrum antimicrobial activity, rapid bactericidal action, and relatively favorable safety profile compared with that of traditional chlorine disinfectants [9–11].
HOCl is a potent oxidizing agent that denatures and aggregates proteins and disrupts cellular membranes by interacting with unsaturated lipid layers [9,10]. In addition, HOCl is endogenously produced by myeloperoxidase in the human immune system and exhibits broad-spectrum antibacterial and antiviral activity [11,12]. HOCl is effective at low concentrations and causes less irritation and material corrosion than sodium hypochlorite (NaOCl), making it suitable for repeated use in healthcare environments [9,11,12]. HOCl has been used in surface disinfection, wound care, food sanitation, and water treatment, with documented efficacy against a wide range of pathogens, and has been approved by the U.S. Food and Drug Administration for these purposes [12].
Recently, the application of HOCl as an aerosol or mist has been proposed as a method for whole-room environmental disinfection, and inhalation of HOCl has been suggested as a therapeutic option for viral infections, such as those due to SARS-CoV-2 [13–15]. Experimental studies using controlled chambers have demonstrated that atomized or dry-mist HOCl can reduce bacterial bioaerosols, including those of P. aeruginosa, in a concentration- and time-dependent manner [10,14,16]. In a study using aerosol chambers (40 × 40 × 100 cm), there was a significant decrease in the survival rate of P. aeruginosa after 30 min of exposure to HOCl compared to that after 3 min of exposure in an atmospheric simulation chamber system [16]. Dry-mist HOCl effectively reduced bacterial and viral populations at 300–500 ppm as well as Bacillus subtilis spore and Aspergillus brasiliensis concentrations at 2000 ppm without damaging medical equipment [10].
However, evidence regarding the effectiveness of HOCl atomization in real hospital rooms against clinically important MDROs, such as CRAB and MRPA, remains limited.
Therefore, the present study aimed to evaluate the bactericidal efficacy of atomized HOCl against CRAB and MRPA in a hospital room. In addition, we compared the effects of atomization with those of direct spraying to assess the potential role of HOCl atomization as an adjunctive environmental disinfection strategy for infection control in healthcare facilities.
CRAB and MRPA were isolated from a transtracheal aspirate sample of a patient (an 88-year-old male) who was admitted to the ICU due to pneumonia and sepsis. Bacterial identification was performed using matrix-assisted laser desorption ionization-time of flight mass spectrometry (bioMérieux), and antimicrobial susceptibility was tested using a VITEK 2 system (bioMérieux) with an AST-N414 gram-negative susceptibility card. A. baumannii showed resistance to all tested antimicrobials except minocycline, whereas P. aeruginosa demonstrated resistance to all antimicrobials except aztreonam. Bacteria were subcultured twice on MacConkey agar (Asan Pharmaceutical) before the experiment. CRAB and MRPA colonies were suspended in 5 and 20 mL of normal saline solution, respectively, to adjust their turbidity to a McFarland turbidity standard of 10. The isolates were prepared using drying and non-drying methods for CRAB and MRPA, respectively. For the drying method, five drops (2 μL each) of the CRAB suspension were placed along a center line drawn on the back of a Petri dish (diameter: 10 cm). The dishes were left in a biological safety cabinet for 2 h to allow complete drying under controlled humidity (~50%). For the non-drying method, 8 mL of the MRPA suspension was poured directly into a Petri dish [17].
This experimental study was conducted in a two-bed hospital room with a volume of 49 m³ at Gyeongsang National University Changwon Hospital, Changwon, Republic of Korea. All experiments were performed with both the air-conditioning and ventilation systems turned off.
The prepared Petri dishes were placed on a bed in the hospital room and the cover was opened before atomization. HOCl (CLFine, Nipro) at concentrations of 40 and 300 ppm and purified water were atomized using ultrasonic humidifiers. Purified water was used as the negative control. Eight ultrasonic atomizers (Seiko Giken, GLeer-100) were used to produce the aerosolized HOCl. A thermo-hygrometer (Sato Keiryoki MFG, SK-L754) data logger was used to monitor the room temperature and humidity. A circulator (Iris Oyama, PCF-HM23) was used to evenly spread the aerosolized HOCl in the room (Fig. 1). All experiments were performed in triplicate. The relative humidity was maintained, approximately, at levels 20% higher than atmospheric humidity. As the atmospheric humidity was approximately 70%, the relative humidity was maintained at approximately 90% throughout the experiment. Increased relative humidity was maintained to ensure stable aerosol dispersion rather than to directly affect bacterial viability. Direct spraying of HOCl solutions (40 and 300 ppm) was performed in the positive controls. Briefly, HOCl was sprayed twice onto the Petri dishes prepared with bacterial suspensions, with a contact time of 5 min. Bacteria were then recovered and cultured as described below. The U.S. Environmental Protection Agency (EPA) requires disinfectant products to be registered with specified contact times on their labels, which must be maintained for effective pathogen inactivation. Contact times for EPA-registered spray disinfectants commonly range from less than one minute to up to ten minutes depending on the formulation and target organisms, and a five-minute contact time falls within this standard range of label-approved contact times for surface disinfectants [18].
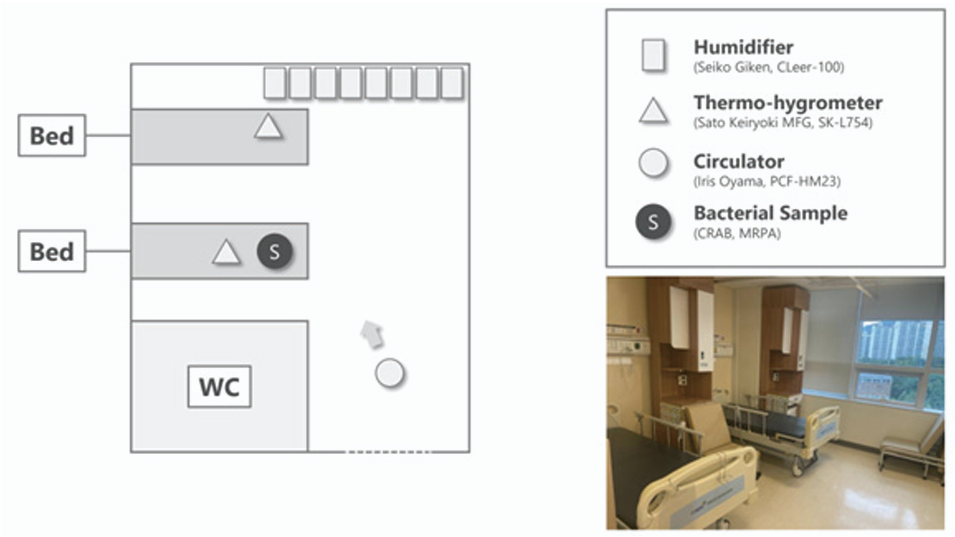
Fig. 1. Schematic representation (left) and real photography (right) of the hospital two-bed room at which the experiment was conducted. Eight humidifiers, two thermo-hygrometers, and one circulator were used for the experiment. The bactericidal effect of atomized HOCl against two bacteria, carbapenemresistant Acinetobacter baumannii (CRAB) and multidrug resistant Pseudomonas aeruginosa (MRPA), was evaluated. HOCl, hypochlorous acid water.
Bacterial samples were collected before atomization and 1, 3, and 5 h after atomization [17]. After adding 2 mL of 0.03% sodium thiosulfate solution (neutralizer) to the CRAB-adhered Petri dishes, the bacteria were scraped off, and 1 mL of this bacteria/neutralizer suspension was placed in a microtube. Regarding MRPA, 1 mL of the bacterial solution in each Petri dish was placed in a microtube. These bacterial solutions were serially diluted 10-fold up to seven times with normal saline. Bacterial dilution (100, 10-1, 10-2, 10-3, 10-4, 10-5, 10-6, and 10-7) samples (100 μL each) were cultured on Soybean Casein Digest agar (Kanto Chemical), and viable colony forming units (CFUs) were determined after incubation for 24 h at 37ºC. For CRAB, the viable bacterial count (CFU) was calculated as follows: measured colony count × dilution factor × 20. For MRPA, the viable bacterial count was calculated as colony count × dilution factor × 80. Bactericidal efficacy was expressed as the logarithmic reduction values (LRVs) or disinfection ratio, which was calculated using the following formula:
Lt = log10 (Mi /Mt )
where:
Lt: Logarithmic reduction value at time point t
Mi: Viable bacterial count before the atomization
Mt: Viable bacterial count after atomization at time point
Atomized HOCl exhibited time- and concentration-dependent bactericidal effects against CRAB and MRPA. At both 40 and 300 ppm, bacterial concentrations progressively decreased over time. Notably, CRAB was eradicated faster than MRPA. Specifically, the times to reach the limit of detection (LOD) — defined as the concentration level at which bacteria are undetectable, that is, 0 CFU is formed— of CRAB and MRPA were 3 and 5 h, respectively, in the presence of HOCl at 40 ppm (Fig. 2). At 300 ppm, CRAB and MRPA were eradicated after 1 and 3 h, respectively. No bacterial inhibition was observed in purified water.
Direct spraying resulted in immediate and pronounced bactericidal effects against both organisms, with higher LRVs observed at earlier time points than with atomization. Both organisms effectively reached their LOD when treated with HOCl at 300 ppm, but variable bactericidal effects were observed when HOCl at 40 ppm was used for treatment (Fig. 3). CRAB was reduced by 2 log when treated with HOCl at 40 ppm and by 5 log in the presence of HOCl 300 ppm, whereas MRPA showed 5–6 log reductions when sprayed with HOCl at 40 and 300 ppm.
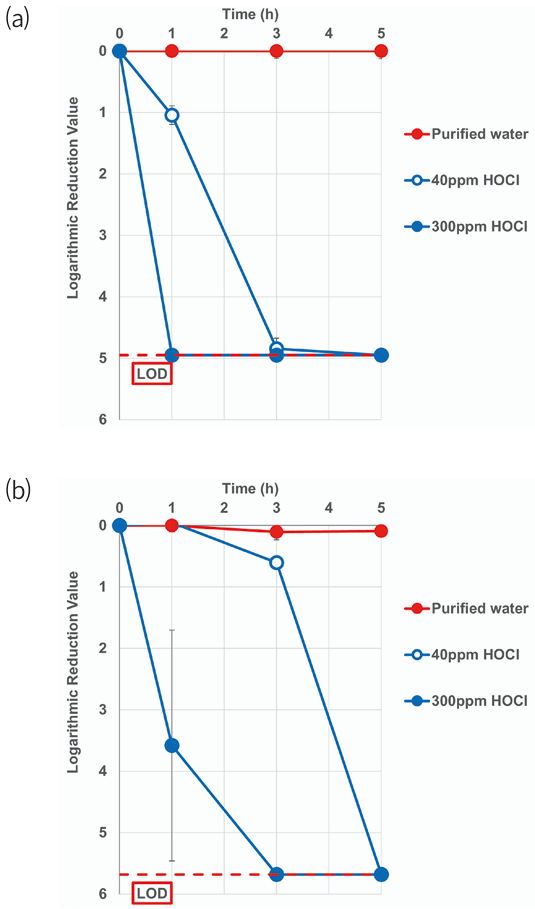
Fig. 2. Logarithmic reduction values of (a) carbapenem-resistant Acinetobacter baumannii (CRAB) and (b) multidrug resistant Pseudomonas aeruginosa (MRPA) after 0, 1, 3, and 5 h of exposure to 40 ppm and 300 ppm of aerosolized HOCl (n = 3, mean ± SD). Purified water was used as negative control. HOCl, hypochlorous acid water; LOD, limit of detection.
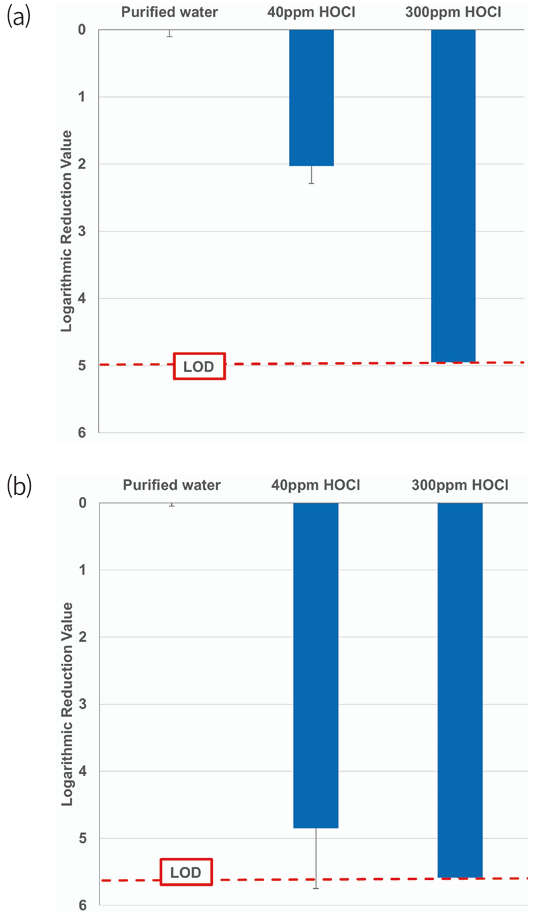
Fig. 3. Logarithmic reduction values of (a) carbapenem-resistant Acinetobacter baumannii (CRAB) and (b) multidrug resistant Pseudomonas aeruginosa (MRPA) after a 5-min exposure to 40 ppm and 300 ppm of sprayed HOCl (n = 3, mean ± SD). Purified water was used as negative control. HOCl, hypochlorous acid water; LOD, limit of detection.
In this study, we demonstrated that atomized HOCl exerted a time-dependent bactericidal effect against CRAB and MRPA in a hospital room with neither air-conditioning nor ventilation. Both tested HOCl concentrations (40 and 300 ppm) produced progressive reductions in viable bacterial counts, with near-complete inactivation observed between 3 and 5 h.
These findings support the effectiveness of mist HOCl exposure in the environmental decontamination of clinically relevant MDROs. Notably, MRPA was eradicated more slowly than CRAB, indicating that more rigorous efforts are required to disinfect MRPA-contaminated areas. The delayed eradication of MRPA compared with that of CRAB may be attributed to the intrinsic biofilm-forming capacity of P. aeruginosa, which is known to reduce susceptibility to disinfectants by limiting penetration and promoting adaptive stress responses [2].
Direct spraying of HOCl resulted in rapid bacterial inactivation, consistent with previous studies demonstrating the strong contact-dependent antimicrobial activity of HOCl [9,11,12]. Compared to the effects seen after atomization, the higher and earlier bactericidal effects were expected after direct spraying, as direct application results in high local disinfectant concentrations. However, it appears that a high HOCl concentration (e.g., 300 ppm) is necessary to reach the bactericidal LOD, when using this direct spraying method.
Overall, HOCl atomization achieved comparable reductions in bacterial populations, highlighting the potential advantage of this approach for whole-room disinfection, where manual surface disinfection may be incomplete or impractical.
The clinical relevance of these findings is underscored by the importance of CRAB and MRPA as the major causes of healthcare-associated infections. CRAB is well known for its ability to persist on dry surfaces for extended periods and contributes to prolonged outbreaks in healthcare settings [3–7]. The demonstrated susceptibility of CRAB to HOCl mist suggests that atomization may help reduce environmental reservoirs that are not adequately addressed by routine cleaning practices.
Similarly, MRPA presents a critical challenge in infection control owing to its intrinsic resistance mechanisms, biofilm-forming capacity, and ability to colonize water-associated environments [1,2]. Numerous studies have identified sinks, drains, and moist surfaces as persistent sources of P. aeruginosa contamination and subsequent patient infection [19]. Previous experimental and in vivo studies have shown that HOCl can effectively inactivate P. aeruginosa on surfaces, in aerosols, and in infected tissues [10,16]. Our findings extend these observations by demonstrating the effectiveness of HOCl atomization against MRPA in a real hospital setting, suggesting the potential role of this approach in infection prevention and control.
The air-conditioning- and ventilation-off conditions applied in this study likely enhanced the disinfection efficacy by minimizing air exchange and allowing sustained exposure of bacteria to HOCl aerosols. Controlled chamber studies have highlighted the importance of stable humidity and aerosol concentrations for effective airborne disinfection [10,14,16], and our results support the applicability of these principles in hospital rooms.
Our study has several limitations. First, only two species of MDROs were evaluated, and the results may not be generalizable to other pathogens. To the best of our knowledge, other MDROs, such as methicillin-resistant Staphylococcus aureus, vancomycin-resistant enterococci, and carbapenem-resistant Klebsiella pneumoniae, have been tested in other countries. We focused on two clinically important gram-negative lactose non-fermenters. In addition, the two different methods (drying and non-drying) applied in this experiment may have interfered with the population decrease rates of the two different bacteria. Second, long-term environmental recolonization after atomization was not assessed. As we performed the bactericidal experiment using a Petri dish, the real bactericidal effects of various types of surfaces or materials in the hospital setting may not be the same. Finally, quantitative statistical comparisons between disinfectant concentrations were limited by the sample size. For the experiment with the two bacteria, 240 Petri dishes were used for each concentration, and this seems to be the maximum number of Petri dishes that can be handled within 5 h.
This study provides strong evidence that atomized HOCl may serve as a useful adjunct to standard infection control measures, particularly in high-risk areas such as ICUs or isolation rooms. If 300 ppm is considered a high concentration of HOCl, 40 ppm could be safely used to eradicate CRAB and MRPA in 3 and 5 h, respectively. From a practical perspective, HOCl atomization may be particularly useful as a terminal room disinfection strategy following the discharge of patients infected or colonized with CRAB or MRPA. This approach may complement routine manual cleaning by targeting residual environmental contaminants in areas that are difficult to disinfect through direct contact. In the future, the bactericidal effects of UV irradiation, glutaraldehyde vapor, and HOCl mist, alone or combined, should be compared and evaluated to maximize healthcare-associated infection control.
This study was conducted as an environmental disinfection efficacy experiment using bacterial isolates, and did not involve human participants or identifiable patient data. This study was approved by the Institutional Review Board (IRB No. NON2024-004) from Gyeongsang National University Changwon Hospital. Informed consent was not required.
No potential conflicts of interest relevant to this article were reported.
This study was supported by the Ministry of Trade, Industry, and Energy of Korea (Grant No. RS 2024-00403563). The funders had no role in the study design, data collection and interpretation, or the decision to submit the manuscript for publication.
The datasets generated during the current study are available from the corresponding author upon request.
The authors express their sincere gratitude to the Nursing Department of Gyeongsang National University Changwon Hospital for the arrangement of the hospital room and their support in this experiment.
1. Reyes J, Komarow L, Chen L, Ge L, Hanson BM, Cober E, et al. Global epidemiology and clinical outcomes of carbapenem-resistant Pseudomonas aeruginosa and associated carbapenemases (POP): a prospective cohort study. Lancet Microbe 2023;4:e159-70.


2. Spagnolo AM, Sartini M, Cristina ML. Pseudomonas aeruginosa in the healthcare facility setting. Rev Med Microbiol 2021;32:169-75.
3. Peleg AY, Seifert H, Paterson DL. Acinetobacter baumannii: emergence of a successful pathogen. Clin Microbiol Rev 2008;21:538-82.


4. Dijkshoorn L, Nemec A, Seifert H. An increasing threat in hospitals: multidrug-resistant Acinetobacter baumannii. Nat Rev Microbiol 2007;5:939-51.

5. Baleivanualala SC, Matanitobua S, Samisoni Y, Soqo V, Smita S, Mailulu J, et al. Environmental contamination with carbapenem-resistant Acinetobacter baumannii in healthcare settings in Fiji: a potential source of infection. Front Cell Infect Microbiol 2024;14:1429443.


6. Fahy SF, O’Connor JA, Lucey B, Sleator RD. Hospital reservoirs of multidrug resistant Acinetobacter species-the elephant in the room! Br J Biomed Sci 2023;80:11098.


7. Wei L, Feng Y, Lin J, Kang X, Zhuang H, Wen H, et al. Handwashing sinks as reservoirs of carbapenem-resistant Acinetobacter baumannii in the intensive care unit: a prospective multicenter study. Front Public Health 2024;12:1468521.


8. Gessi A, Formaglio P, Semeraro B, Summa D, Tamisari E, Tamburini E. Electrolyzed hypochlorous acid (HOCl) aqueous solution as low-impact and eco-friendly agent for floor cleaning and sanitation. Int J Environ Res Public Health 2023;20:6712.


9. Block MS and Rowan BG. Hypochlorous acid: a review. J Oral Maxillofac Surg 2020;78:1461-6.


10. Nasiłowska B, Włodarski M, Kaliszewski M, Bogdanowicz Z, Krzowski Ł, Kopczyński K, et al. Decontamination effect of hypochlorous acid dry mist on selected bacteria, viruses, spores, and fungi as well as on components of electronic systems. Int J Mol Sci 2024;25:7198.


11. Yan P, Daliri EBM, Oh DH. New clinical applications of electrolyzed water: a review. Microorganisms 2021;9:136.


12. Boecker D, Zhang Z, Breves R, Herth F, Kramer A, Bulitta C. Antimicrobial efficacy, mode of action and in vivo use of hypochlorous acid (HOCl) for prevention or therapeutic support of infections. GMS Hyg Infect Control 2023;18:Doc07.
13. Winter M, Boecker D, Posch W. Hypochlorous acid (HOCl) as a promising respiratory antiseptic. Viruses 2025;17:1219.


14. Park J, Lee KH, Song YG, Park H, Lee KS. Development of optimal indoor air disinfection and ventilation protocols for airborne infectious diseases. PLoS One 2024;19:e0311274.


15. Rasmussen E, Robins LI, Stone J, Williams JF. Inhalation of microaerosolized hypochlorous acid (HOCl): biochemical, antimicrobial, and pathological assessment. Arch Intern Med Res 2022;5:311-8.
16. Norkaew T, Narikawa S, Nagashima U, Uemura R, Noda J. Efficacy of treating bacterial bioaerosols with weakly acidic hypochlorous water: a simulation chamber study. Heliyon 2024;10:e26574.


17. Takahashi M and Simooki O. Evaluation of test methods and the efficacy of hypochlorous acid water atomization against Pseudomonas aeruginosa in hospital rooms. Infect Dis Health 2025;31:100394.

18. U.S. Environmental Protection Agency. Selected EPA-registered disinfectants – contact time and product label guidance. https://www.epa.gov/pesticide-registration/selected-epa-registered-disinfectants [Online] (last visited on 24 February 2026).
19. Chia PY, Sengupta S, Kukreja A, S L Ponnampalavanar S, Ng OT, Marimuthu K. The role of hospital environment in transmissions of multidrug-resistant gram-negative organisms. Antimicrob Resist Infect Control 2020;9:29.


1. Reyes J, Komarow L, Chen L, Ge L, Hanson BM, Cober E, et al. Global epidemiology and clinical outcomes of carbapenem-resistant Pseudomonas aeruginosa and associated carbapenemases (POP): a prospective cohort study. Lancet Microbe 2023;4:e159-70.


2. Spagnolo AM, Sartini M, Cristina ML. Pseudomonas aeruginosa in the healthcare facility setting. Rev Med Microbiol 2021;32:169-75.
3. Peleg AY, Seifert H, Paterson DL. Acinetobacter baumannii: emergence of a successful pathogen. Clin Microbiol Rev 2008;21:538-82.


4. Dijkshoorn L, Nemec A, Seifert H. An increasing threat in hospitals: multidrug-resistant Acinetobacter baumannii. Nat Rev Microbiol 2007;5:939-51.

5. Baleivanualala SC, Matanitobua S, Samisoni Y, Soqo V, Smita S, Mailulu J, et al. Environmental contamination with carbapenem-resistant Acinetobacter baumannii in healthcare settings in Fiji: a potential source of infection. Front Cell Infect Microbiol 2024;14:1429443.


6. Fahy SF, O’Connor JA, Lucey B, Sleator RD. Hospital reservoirs of multidrug resistant Acinetobacter species-the elephant in the room! Br J Biomed Sci 2023;80:11098.


7. Wei L, Feng Y, Lin J, Kang X, Zhuang H, Wen H, et al. Handwashing sinks as reservoirs of carbapenem-resistant Acinetobacter baumannii in the intensive care unit: a prospective multicenter study. Front Public Health 2024;12:1468521.


8. Gessi A, Formaglio P, Semeraro B, Summa D, Tamisari E, Tamburini E. Electrolyzed hypochlorous acid (HOCl) aqueous solution as low-impact and eco-friendly agent for floor cleaning and sanitation. Int J Environ Res Public Health 2023;20:6712.


9. Block MS and Rowan BG. Hypochlorous acid: a review. J Oral Maxillofac Surg 2020;78:1461-6.


10. Nasiłowska B, Włodarski M, Kaliszewski M, Bogdanowicz Z, Krzowski Ł, Kopczyński K, et al. Decontamination effect of hypochlorous acid dry mist on selected bacteria, viruses, spores, and fungi as well as on components of electronic systems. Int J Mol Sci 2024;25:7198.


11. Yan P, Daliri EBM, Oh DH. New clinical applications of electrolyzed water: a review. Microorganisms 2021;9:136.


12. Boecker D, Zhang Z, Breves R, Herth F, Kramer A, Bulitta C. Antimicrobial efficacy, mode of action and in vivo use of hypochlorous acid (HOCl) for prevention or therapeutic support of infections. GMS Hyg Infect Control 2023;18:Doc07.
13. Winter M, Boecker D, Posch W. Hypochlorous acid (HOCl) as a promising respiratory antiseptic. Viruses 2025;17:1219.


14. Park J, Lee KH, Song YG, Park H, Lee KS. Development of optimal indoor air disinfection and ventilation protocols for airborne infectious diseases. PLoS One 2024;19:e0311274.


15. Rasmussen E, Robins LI, Stone J, Williams JF. Inhalation of microaerosolized hypochlorous acid (HOCl): biochemical, antimicrobial, and pathological assessment. Arch Intern Med Res 2022;5:311-8.
16. Norkaew T, Narikawa S, Nagashima U, Uemura R, Noda J. Efficacy of treating bacterial bioaerosols with weakly acidic hypochlorous water: a simulation chamber study. Heliyon 2024;10:e26574.


17. Takahashi M and Simooki O. Evaluation of test methods and the efficacy of hypochlorous acid water atomization against Pseudomonas aeruginosa in hospital rooms. Infect Dis Health 2025;31:100394.

18. U.S. Environmental Protection Agency. Selected EPA-registered disinfectants – contact time and product label guidance. https://www.epa.gov/pesticide-registration/selected-epa-registered-disinfectants [Online] (last visited on 24 February 2026).
19. Chia PY, Sengupta S, Kukreja A, S L Ponnampalavanar S, Ng OT, Marimuthu K. The role of hospital environment in transmissions of multidrug-resistant gram-negative organisms. Antimicrob Resist Infect Control 2020;9:29.

