1Department of Laboratory Medicine, 2Department of Ophthalmology, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea
Corresponding to Eun Jeong Won, E-mail: ejwon@amc.seoul.kr
Ann Clin Microbiol 2024;27(2):149-153. https://doi.org/10.5145/ACM.2024.27.2.9
Received on 19 April 2024, Revised on 20 May 2024, Accepted on 20 May 2024, Published on 20 June 2024.
Copyright © Korean Society of Clinical Microbiology.
This is an Open Access article which is freely available under the Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License (CC BY-NC-ND) (https://creativecommons.org/licenses/by-nc-nd/4.0/).
Acanthamoeba species are ubiquitous, free-living organisms found in the environment. They can cause a sight-threatening cornea disease, termed Acanthamoeba keratitis, and are often misdiagnosed, causing delayed administration of the correct treatment. Herein, we report a case of Acanthamoeba keratitis diagnosed without culture. A 12-year-old girl with a history of wearing contact lenses presented with complaints of pain, irritation, and hyperemia in the left eye. Corneal scraping-smeared slide, and liquids with contact lenses were submitted to the clinical microbiology laboratory. Cultures of Acanthamoeba spp. were not available; thus, they were stained with calcofluor white. The isolation of Acanthamoeba from the corneal scraping allowed the detection of trophozoites and cysts based on their morphological characteristics. PCR targeting the 18s rRNA gene and subsequent sequencing revealed 99% identity with the Acanthamoeba spp. Although it is challenging to find real-world evidence of Acanthamoeba in clinical microbiology without using culture methods, this case underscores the need for clinical microbiology laboratories to maintain their inspection capabilities.
Acanthamoeba keratitis, cyst, trophozoite, morphology
Acanthamoeba species are ubiquitous, free-living organisms found in the environment. They can cause a sight-threatening cornea disease, termed Acanthamoeba keratitis, and are often misdiagnosed, causing delayed treatment. Although the gold standard diagnosis is amoeba isolation from corneal scraping samples, it is challenging to find evidence of Acanthamoeba in real-world clinical microbiology practice. Herein, we report a case of Acanthamoeba keratitis diagnosed without culture, and describe several diagnostic methods for Acanthamoeba that can be useful in actual clinical microbiology laboratories.
Patient information: A 12 year-old girl with a history of wearing contact lenses presented with pain, irritation, and hyperemia in the left eye visited the ophthalmology clinic at Asan Medical Center, South Korea.
Clinical findings: During slit-lamp microscopic examination of the patient’s left eye, radial keratoneuritis was observed in the central corneal stroma, whereas the vitreous and retina showed no abnormalities (Fig. 1).
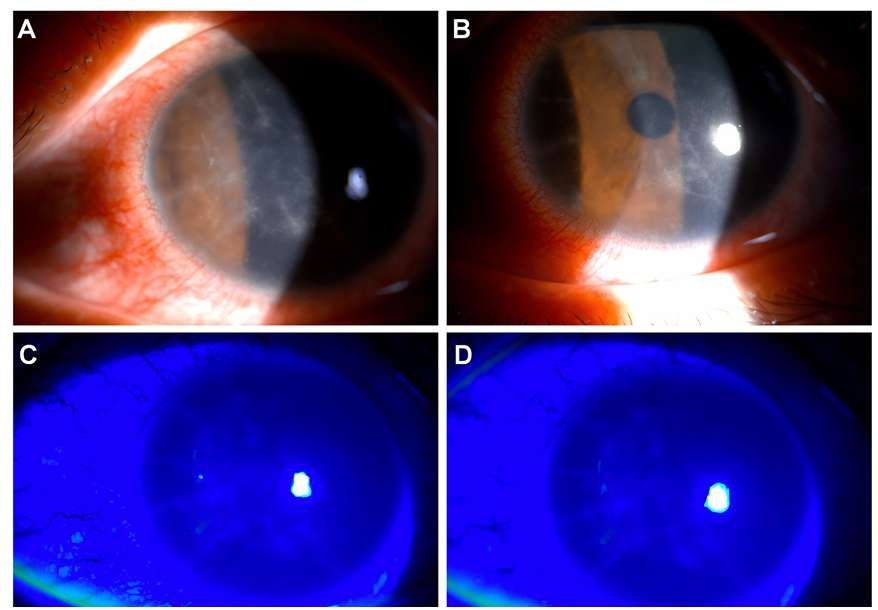
Fig. 1. Anterior segment photographs of the patient in this case study. The top two anterior segment images were taken under slit-lamp microscope (A, B) and the bottom two images were under cobalt blue light after fluorescein dye staining (C, D). During the slit-lamp microscopy examination of the patient’s left eye, radial keratoneuritis was observed in the central corneal stroma, while vitreous and retina showed no abnormalities.
Diagnostic assessment: Corneal scraping was performed, and a direct smear slide was prepared. The slide and liquids with contact lenses were submitted to a clinical microbiology laboratory. Cultures for Acanthamoeba spp. were not available; thus, they were stained with calcofluor white as previously described [1]. Isolation of Acanthamoeba spp. from the corneal scraping and liquids allowed the detection of trophozoites and cysts under both light and fluorescence microscopes (Fig. 2). The Acanthamoeba cysts were approximately 10–15 μm in diameter, round, with a plica-like surface (Fig. 2A, 2C). The viable cysts were round or oval with double cyst walls, that is, ectocyst and endocyst walls. The ectocysts contracted into wrinkles and were separated from the thin and smooth endocysts. The trophozoites (about 20–25 μm in diameter) were pleomorphic and exhibited large lamellipodium of varying size and shape. A single large nucleus and numerous vacuoles with diverse contents in trophozoites were clearly observed by light microscopy but were unremarkable by fluorescent microscopy (Fig. 2B, 2D). The microscopic morphology was consistent with Acanthamoeba spp. reported to the ophthalmology department. To molecularly identify the microorganisms, a segment of the 18 small subunit ribosomal ribonucleic acid (rRNA) gene was amplified. Genomic DNA was extracted from the samples using the QIAamp DNA Mini Kit (Qiagen) according to the manufacturer’s instructions. Subsequently, polymerase chain reaction (PCR) was conducted employing the primer set JDP1 (5′-GGCCCAGATCGTTTACCGTGAA-3′) and JDP2 (5′-TCTCACAAG CTGCTAGGGAGTCA-3′), as documented in the literature [2]. Briefly, the PCR process involved an initial incubation at 94°C for 5 min, followed by 35 cycles of denaturation at 94°C for 1 min, primer annealing at 55°C for 1 min, and elongation at 72°C for 1 min, with a final elongation step at 72°C for 10 min to complete the amplification process. The PCR product, 436 base pairs in length, was sent to Cosmo Genetech for direct sequencing. The chromatograms of sequences were trimmed manually and assembled using the SeqMan software (DNASTAR). Basic local alignment search tool searches of the assembled sequences revealed 99% identity with Acanthamoeba spp. (GenBank accession no. MG825460.1) and conclusively identified the microorganism as Acanthamoeba spp.
Therapeutic intervention: Based on the morphological characteristics observed under the microscope, the patient was prescribed 0.02% polyhexamethylene biguanide eyedrops as a treatment on the day of the visit to our hospital.
Follow-up and outcomes: The patient underwent five months of pharmacological treatment, and the initially observed radial keratoneuritis showed significant improvement.
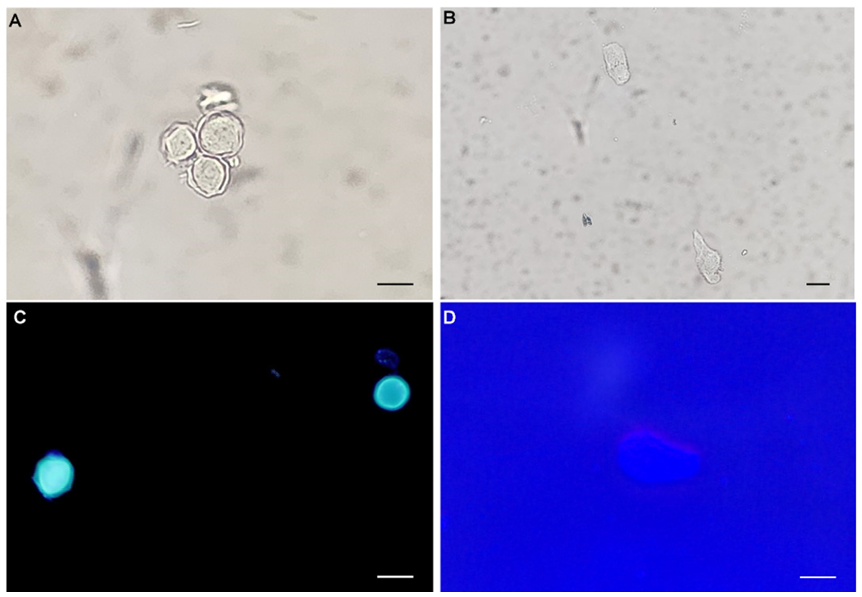
Fig. 2. Morphological analysis of Acanthamoeba spp. using calcofluor-white staining. (A) Acanthamoeba cysts in a group under light microscopy (400×, scale bar = 10 μm). (B) Acanthamoeba trophozoites with a single large nucleus and numerous vacuoles were conspicuous under light microscopy (200×, scale bar = 10 μm). (C) Remarkable Acanthamoeba cysts with double cyst walls under fluorescence microscopy (400×, scale bar = 10 μm). (D) Acanthamoeba trophozoites were relatively unremarkable under fluorescence microscopy (400×, scale bar = 10 μm).
Since the first report of Acanthamoeba keratitis in 1975, its incidence has increased, particularly among contact lens users [3]. In Korea, most published reports on the clinical patterns and treatments of Acanthamoeba keratitis have been case reports [4,5]; however, the incidence is perceived to be low, and the exact prevalence remains unclear [6]. The gold standard for diagnosing Acanthamoeba keratitis involves scraping the corneal epithelium, particularly from areas with severe epithelial damage, and culturing on Escherichia coli-seeded non-nutrient agar plates. This method allows the observation of Acanthamoeba predation paths on E. coli, demonstrating a detection rate of 60%–76% [7]. Several additional tests have been suggested to ascertain the growth rate, temperature tolerance, or pathogenicity of isolated amoebae, and axenization using proteose peptone-yeast extract-glucose medium is required [8]. Nonetheless, this culture method is time-consuming, taking a 1 week or more, which delays diagnosis and treatment. Although culture has been recognized as the gold standard for Acanthamoeba laboratory diagnosis [1], it has not been well established in clinical laboratories. Instead of culture, several staining methods, such as lactophenol-cotton blue, acridine orange, calcofluor white, silver, and hematoxylin and eosin, can be useful for the identification of Acanthamoeba spp. from clinical specimens [1,9]. In this study, we found that Acanthamoeba cysts appeared light green on staining with calcofluor white. Direct microscopy of the original sample can detect characteristic cysts and trophozoites [1]. In this case, the causative agent was diagnosed through observation of Acanthamoeba morphology via a direct smear of the original sample without performing an axenic culture. However, relying solely on morphology for Acanthamoeba keratitis diagnosis through microscopic examination can lead to a misdiagnosis without experienced inspectors. This necessitates quality control in clinical laboratories, and continuous education regarding this uncommon pathogen. In addition to direct observation, we performed PCR using JDP1 and JDP2 primer sets, as previously described. The PCR method is well-established for detecting the 18S rRNA gene, facilitating genotyping in most cases through sequencing [2]. In addition to sequencing, real-time PCR has been preferred in clinical laboratories because there is no need for postamplification handling, leading to faster analysis and reduced risk of amplicon contamination [10]. Several real-time PCR-based assays have been developed and evaluated [11], but no commercial real-time PCR kits for the diagnosis of Acanthamoeba keratitis are available in Korea. Financial and procedural hurdles due to the non-reimbursable status of axenic cultures and PCR in certain diagnostic frameworks should be further overcome. Although it is challenging to find real-world evidence of Acanthamoeba in clinical microbiology without using culture methods, this case underscores the need for clinical microbiology laboratories to maintain their inspection capabilities. Furthermore, this study highlights the need for insights into Acanthamoeba diagnosis in clinical laboratories using various diagnostic tools.
The study was approved by the Institutional Review Board of Asan Medical Center (No. 2024-0519) and the requirement for informed consent was waived because of the retrospective nature of the study.
Eun Jeong Won has been an associate editor of the Annals of Clinical Microbiology since January 2024. However, she was not involved in the review process of this article. No other potential conflict of interest relevant to this article was reported.
This study was supported by grants from the National Research Foundation of Korea (NRF) funded by the Ministry of Education, Science, and Technology (Grant No. 2022R1C1C1002741).
1. Lorenzo-Morales J, Khan NA, Walochnik J. An update on Acanthamoeba keratitis: diagnosis, pathogenesis and treatment. Parasite 2015;22:10. 


2. Schroeder JM, Booton GC, Hay J, Niszl IA, Seal DV, Markus MB, et al. Use of subgenic 18S ribosomal DNA PCR and sequencing for genus and genotype identification of Acanthamoebae from humans with keratitis and from sewage sludge. J Clin Microbiol 2001;39:1903–11. 


3. Jones BR, McGill JI, Steele AD. Recurrent suppurative kerato-uveitis with loss of eye due to infection by Acanthamoeba castellani. Trans Ophthalmol Soc U K 1975;95:210–3.
4. Lee JE, Hahn TW, Oum BS, Choi HY, Yu HS, Lee JS. Acanthamoeba keratitis related to orthokeratology. Int Ophthalmol 2007;27:45–9. 

5. Lee JS, Hahn TW, Choi SH, Yu HS, Lee JE. Acanthamoeba keratitis related to cosmetic contact lenses. Clin Exp Ophthalmol 2007;35:775–7. 

6. Kong HH, Shin JY, Yu HS, Kim J, Hahn TW, Hahn YH, et al. Mitochondrial DNA restriction fragment length polymorphism (RFLP) and 18S small-subunit ribosomal DNA PCRRFLP analyses of Acanthamoeba isolated from contact lens storage cases of residents in southwestern Korea. J Clin Microbiol 2002;40:1199–206. 


7. Yoon HY and Jeon HS. Diagnosis and management of Acanthamoeba keratitis. Ann Optom Contact Lens 2022;21:104–8. 
8. Chan LL, Mak JW, Low YT, Koh TT, Ithoi I, Mohamed SM. Isolation and characterization of Acanthamoeba spp. from air-conditioners in Kuala Lumpur, Malaysia. Acta Trop 2011;117:23–30. 


9. Thomas PA and Kuriakose T. Rapid detection of Acanthamoeba cysts in corneal scrapings by lactophenol cotton blue staining. Arch Ophthalmol 1990;108:168. 

10. Mackay IM. Real-time PCR in the microbiology laboratory. Clin Microbiol Infect 2004;10:190-212. 

11. Qvarnstrom Y, Visvesvara GS, Sriram R, da Silva AJ. Multiplex real-time PCR assay for simultaneous detection of Acanthamoeba spp., Balamuthia mandrillaris, and Naegleria fowleri. J Clin Microbiol 2006;44:3589–95. 


1. Lorenzo-Morales J, Khan NA, Walochnik J. An update on Acanthamoeba keratitis: diagnosis, pathogenesis and treatment. Parasite 2015;22:10.
2. Schroeder JM, Booton GC, Hay J, Niszl IA, Seal DV, Markus MB, et al. Use of subgenic 18S ribosomal DNA PCR and sequencing for genus and genotype identification of Acanthamoebae from humans with keratitis and from sewage sludge. J Clin Microbiol 2001;39:1903–11.
3. Jones BR, McGill JI, Steele AD. Recurrent suppurative kerato-uveitis with loss of eye due to infection by Acanthamoeba castellani. Trans Ophthalmol Soc U K 1975;95:210–3.
4. Lee JE, Hahn TW, Oum BS, Choi HY, Yu HS, Lee JS. Acanthamoeba keratitis related to orthokeratology. Int Ophthalmol 2007;27:45–9.
5. Lee JS, Hahn TW, Choi SH, Yu HS, Lee JE. Acanthamoeba keratitis related to cosmetic contact lenses. Clin Exp Ophthalmol 2007;35:775–7.
6. Kong HH, Shin JY, Yu HS, Kim J, Hahn TW, Hahn YH, et al. Mitochondrial DNA restriction fragment length polymorphism (RFLP) and 18S small-subunit ribosomal DNA PCRRFLP analyses of Acanthamoeba isolated from contact lens storage cases of residents in southwestern Korea. J Clin Microbiol 2002;40:1199–206.
7. Yoon HY and Jeon HS. Diagnosis and management of Acanthamoeba keratitis. Ann Optom Contact Lens 2022;21:104–8.
8. Chan LL, Mak JW, Low YT, Koh TT, Ithoi I, Mohamed SM. Isolation and characterization of Acanthamoeba spp. from air-conditioners in Kuala Lumpur, Malaysia. Acta Trop 2011;117:23–30.
9. Thomas PA and Kuriakose T. Rapid detection of Acanthamoeba cysts in corneal scrapings by lactophenol cotton blue staining. Arch Ophthalmol 1990;108:168.
10. Mackay IM. Real-time PCR in the microbiology laboratory. Clin Microbiol Infect 2004;10:190-212.
11. Qvarnstrom Y, Visvesvara GS, Sriram R, da Silva AJ. Multiplex real-time PCR assay for simultaneous detection of Acanthamoeba spp., Balamuthia mandrillaris, and Naegleria fowleri. J Clin Microbiol 2006;44:3589–95.